Application of the LAMBDA MINIFOR in a Rumen Membrane Bioreactor for VFA Production
The global abundance of lignocellulosic biomass (LCBM)—such as crop residues—represents a vast and underutilized carbon resource. However, its recalcitrant structure demands a highly specialized microbial community for effective degradation. Ruminant animals achieve this naturally in their rumen, where cellulolytic and fermentative microorganisms convert LCBM into volatile fatty acids (VFAs)—the primary energy source for the host.
To reproduce this highly efficient digestion process in vitro, researchers from from the University of Technology Sydney developed a rumen membrane bioreactor (rumen-MBR). At the core of this system, the 3 L LAMBDA MINIFOR bioreactor played a critical role. Its flexible configuration, precise environmental control, and modular design enabled successful simulation of rumen-like conditions and continuous VFA extraction using a submerged ultrafiltration (UF) membrane.
Reference: Nguyen AQ, Nguyen LN, Johir MAH, Ngo H-H, Chaves AV, Nghiem LD. Derivation of volatile fatty acid from crop residues digestion using a rumen membrane bioreactor: A feasibility study. Bioresour Technol. 2020;317:123571. doi:10.1016/j.biortech.2020.123571
A LAMBDA MINIFOR Bioreactor 3L was used for the rumen membrane bioreactor (rumen-MBR). The system was configured with two peristaltic pumps for continuous feeding and permeate extraction, stirring by biomimicking up-down agitation, and a multi-parameter probe capable of monitoring redox potential, temperature, and pH.
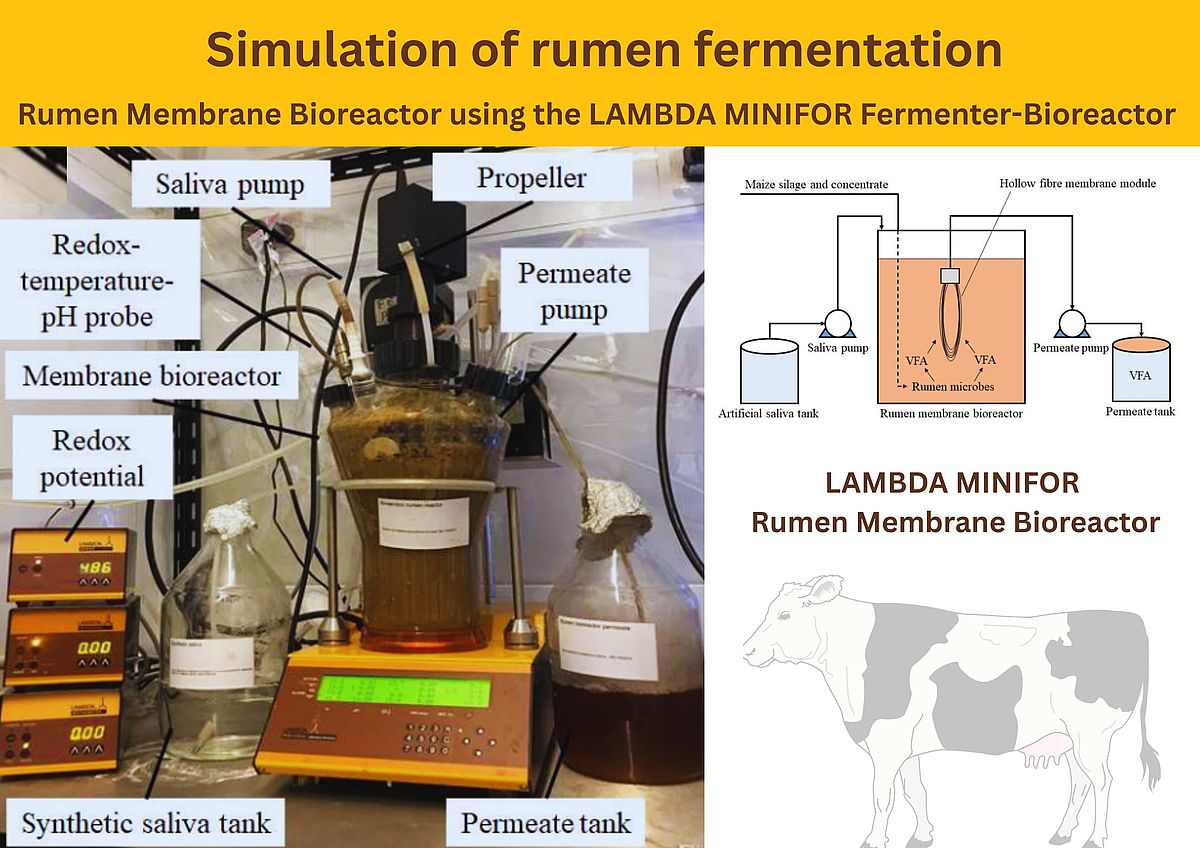
Image Credit and Reference: Nguyen, Q. A. (2022). Microbial community analysis using next-generation sequencing (NGS) for rumen-based anaerobic digestion systems. Doctoral thesis, University of Technology Sydney. (Chapter 6: Derivation of volatile fatty acid from crop residues digestion using a rumen membrane bioreactor)
https://opus.lib.uts.edu.au/bitstream/10453/164384/2/02whole.pdf
Temperature regulation was achieved through the bioreactor's Infrared heating which offers gentle and precise temperature regulation. In addition, a submerged hollow-fiber membrane unit was installed within the vessel. This membrane module was fabricated using epoxy resin to embed 20 polyvinylidene difluoride (PVDF) fibers each measuring 30 cm in length with a 0.04 µm pore size, yielding an effective filtration surface area of approximately 0.02 m².
For reactor start-up, 1 L of rumen fluid was mixed with 1 L of modified McDougall’s saliva, together with 20 g of a maize silage–concentrate mixture (60:40 w/w). The reactor was sparged with pure nitrogen gas until the oxidation–reduction potential decreased to below –300 mV, establishing anaerobic conditions representative of the natural rumen. The inoculated reactor contents were maintained under continuous mixing at 150 rpm (corresponds to 2.5 Hz) using a three-bladed impeller, while the temperature was controlled at 39 °C to emulate in vivo rumen conditions.
Continuous operation was achieved by simultaneously running the feed and permeate pumps to maintain a hydraulic retention time (HRT) of 57 hours, corresponding to an operational permeate flux of 2.07 L/m²·h. This flux was intentionally kept low to reduce the likelihood of membrane fouling. Each day, 100 mL of mixed liquor was removed and replaced with a fresh mixture containing 2 g of substrate in 100 mL of saliva, resulting in an organic loading rate of 1 g/L·day (equivalent to ~1.14 kg COD/m³·day). This moderate loading regime helped prevent insufficient mixing and avoided overloading of the microbial community. Under these conditions, the solids retention time (SRT) of the rumen-MBR was calculated to be 480 hours.
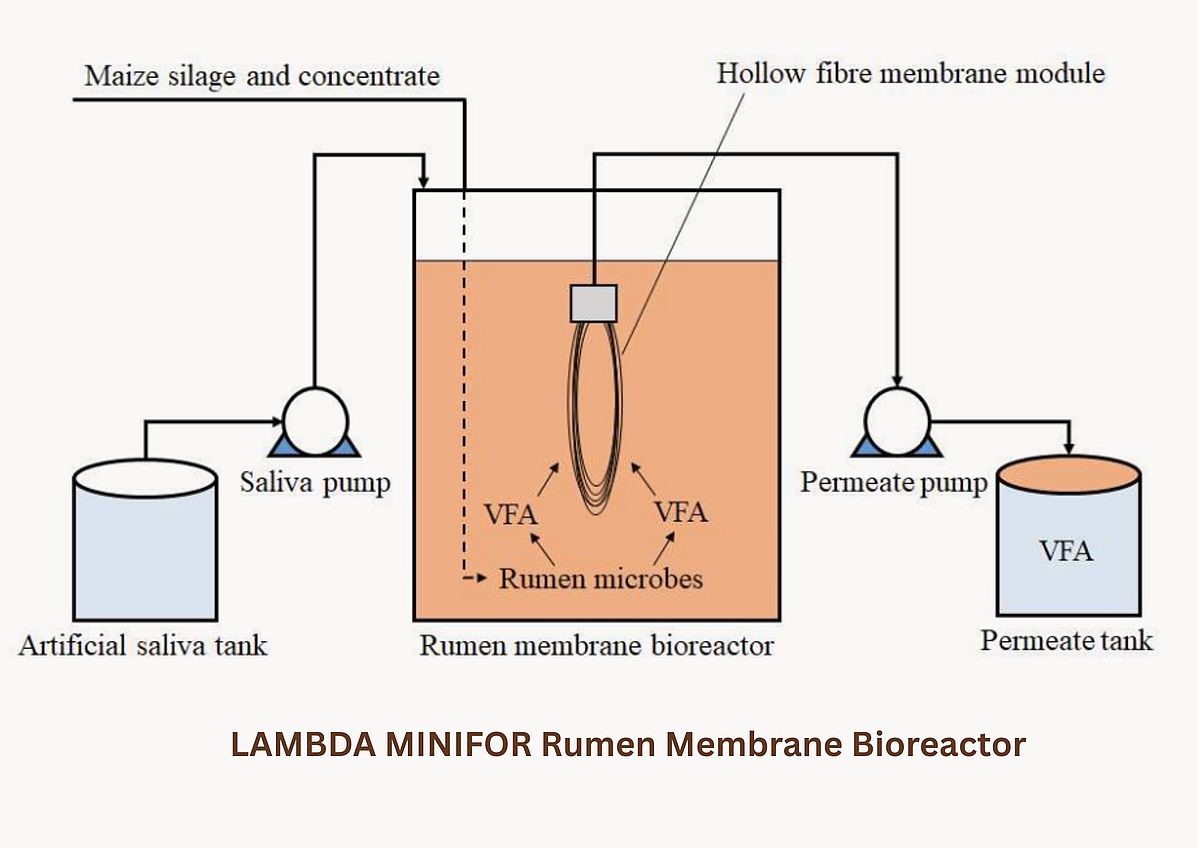
Image Credit and Reference: Nguyen, Q. A. (2022). Microbial community analysis using next-generation sequencing (NGS) for rumen-based anaerobic digestion systems. Doctoral thesis, University of Technology Sydney. (Chapter 6: Derivation of volatile fatty acid from crop residues digestion using a rumen membrane bioreactor) https://opus.lib.uts.edu.au/bitstream/10453/164384/2/02whole.pdf
The system was operated for 44 days. During this period, the pH and biogas production were monitored continuously, while analyses of volatile fatty acids (VFAs), total solids (TS), volatile solids (VS), and soluble chemical oxygen demand (sCOD) were conducted twice weekly to assess reactor performance and stability.
Did You Know? LAMBDA MINIFOR Supports Dual-flow continuous culture fermenter or RUSITEC - Rumen Simulation Technique:
The Minifor Fermenter system features a customized dual-flow design, with a side arm that can be connected to a tube for directing overflow into a collection container.
Why the LAMBDA MINIFOR was chosen for the Rumen Bioreactor?
Rumen digestion depends on a stable temperature, strict anaerobic conditions, rhythmic mixing, and the presence of a microbial community. These requirements guided the choice of reactor, and the MINIFOR offered several advantages:
Reliable environmental control
The natural rumen operates at around 39 °C, and the MINIFOR’s integrated heating system held this temperature consistently throughout the 44-day experiment. The vessel’s ports allowed purging with nitrogen gas until the redox potential dropped below –300 mV, establishing the necessary anaerobic environment for rumen microorganisms.
The MINIFOR Fermenter-Bioreactor is equipped with MASSFLOW Gas Regulator for regulated supply of Air or Nitrogen.
Integrated monitoring
The system was equipped with a combined pH, redox, and temperature probe, enabling continuous measurement of the three most important process parameters. Maintaining a stable pH is essential in rumen studies because VFA accumulation can quickly cause acidification. The MINIFOR’s real-time monitoring helped prevent such drops and ensured steady operation.
Exit-gas analysis for RUSITEC rumen simulation.
Integration with the LAMBDA METHAMETER and LAMBDA CARBOMETER enables continuous monitoring of methane and CO₂ in the exhaust gas, supporting detailed evaluation of ruminal fermentation and gas production in RUSITEC systems.
Suitable agitation
Inside the reactor, the microbial community and fibrous substrate were mixed using the MINIFOR’s overhead three-silicone bladed impeller, operated at 150 rpm. This agitation speed was strong enough to keep the lignocellulosic material suspended but gentle enough to preserve the structure of the rumen microbes—conditions that mirror natural rumen motility.
Flexibility for membrane integration
A key part of the study was the use of a submerged ultrafiltration membrane module. The MINIFOR headplate and side ports provided enough space to install:
-
a PVDF hollow-fiber membrane bundle (20 fibers, 30 cm length, 0.04 µm pore size),
-
a permeate pump for continuous VFA withdrawal, and
-
a feed pump for artificial saliva addition.
This modularity made it straightforward to combine fermentation and membrane separation in a single vessel.
Summary
The study demonstrates how the LAMBDA MINIFOR Fermenter-Bioreactor can be adapted for complex anaerobic and membrane-coupled processes. Its precise temperature control, dependable agitation, integrated sensing, and flexible design made it possible to reproduce core rumen functions on a laboratory scale. The success of the rumen-MBR underscores the MINIFOR’s suitability for: Rumen membrane bioreactor, to simulate the rumen microbiome, RUSITEC model, Dual-flow continuous culture system.
Ready to elevate your research? Explore the LAMBDA Minifor2Bio touch Fermenter-Bioreactor today and discover how it can transform your work. You can read more at the product page - www.lambda-instruments.com/fermenter-bioreactor-touch/
NEW! - The new Minifor2Bio touch laboratory fermenters-bioreactors are now available!
Read more: https://fermenters.eu/
If you have any inquiries, require a quotation, or need pricing information for our fermentor-bioreactor system, feel free to reach out to us at sales@lambda-instruments.com.
