Application of the LAMBDA OMNICOLL Fraction Collector in Hexavalent Chromium Recovery Research
Hexavalent chromium (Cr(VI)) is one of the most hazardous contaminants found in industrial wastewater, particularly in effluents generated by chromium production plants. Because of its high toxicity and environmental impact, industries must ensure that chromium concentrations in wastewater are reduced to extremely low levels before discharge.
A research study titled “The Regeneration of Saturated Ionites after Extraction of Hexavalent Chromium from Wastewater of Chromium Production Plant” investigated the recovery of chromium from wastewater using anion exchange resins and evaluated different regeneration methods. In this work, the LAMBDA OMNICOLL Fraction Collector was used to automatically collect column effluent samples during ion exchange and regeneration experiments, enabling accurate monitoring of chromium concentration throughout the process.
Reference: Pastukhov, A. M., Chernyi, M. L., & Skripchenko, S. Yu. (2019). The regeneration of saturated ionites after extraction of hexavalent chromium from wastewater of chromium production plant. AIP Conference Proceedings, 2174, 020047. doi.org/10.1063/1.5134198
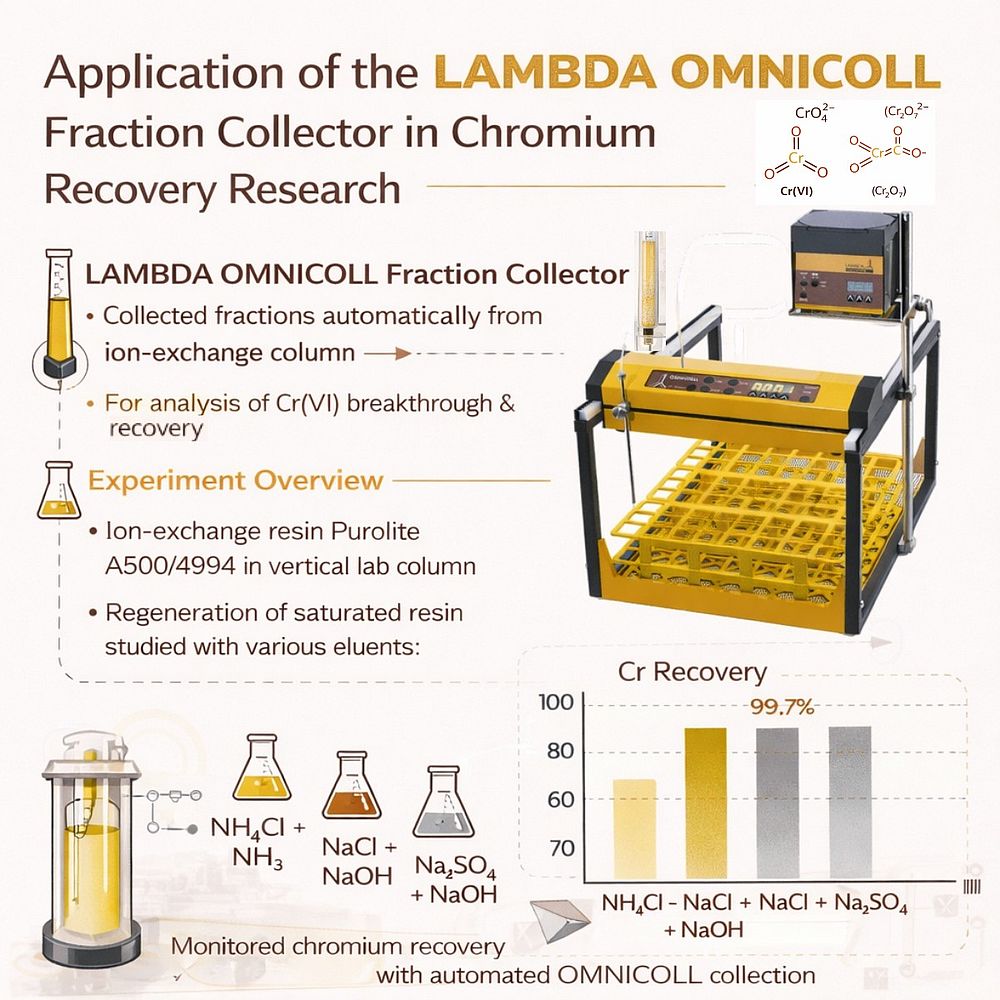
Figure 1: Pictorial representation of work overview
Ion Exchange Removal of Hexavalent Chromium
The study focused on removing hexavalent chromium from aqueous solutions using the strongly basic anion exchange resin Purolite A500/4994. Ion exchange is considered one of the most promising technologies for treating chromium-containing wastewater because it can reduce chromium concentrations to very low levels while allowing recovery of valuable chromium compounds.
In the experimental setup, model chromium-containing solutions were prepared with a chromium concentration of approximately 1.834 g/L and sulfate concentration of 2 g/L. The pH of the solution was adjusted to pH 7 before being introduced into the ion exchange column.
The resin was packed into a vertical laboratory column, and the chromium solution was continuously pumped through the column using a peristaltic pump at a constant flow rate of approximately five column volumes per hour. During this process, chromium ions were adsorbed by the anion exchange resin until the resin became saturated.
Role of the LAMBDA OMNICOLL Fraction Collector
During the column experiments, the outlet solution from the ion exchange column needed to be collected in sequential samples in order to monitor the adsorption process. The LAMBDA OMNICOLL Fraction Collector was used to automatically collect the effluent fractions at regular intervals.
This automated fraction collection ensured consistent sampling during the entire experiment. The collected fractions were later analyzed using a titrimetric method with a standard iron(II) solution to determine the chromium concentration in each fraction.
By analyzing the chromium concentration in the collected fractions, it was possible to monitor the breakthrough behavior of chromium in the column and determine when the resin became saturated. Automated sampling also helped maintain reproducibility and minimized the risk of human error during long experimental runs.
Investigation of Resin Regeneration
After the ion exchange resin reached saturation, the next stage of the research focused on regenerating the resin and recovering the adsorbed chromium. The desorption process was carried out in dynamic column conditions using several different regeneration solutions.
The eluates leaving the column during the regeneration stage were again collected using the LAMBDA OMNICOLL Fraction Collector. These fractions allowed precise monitoring of the chromium desorption process and helped determine the effectiveness of different eluents.
Three main regeneration solutions were tested:
ammonium chloride solutions with added ammonia
sodium chloride solutions with sodium hydroxide
sodium sulfate solutions with sodium hydroxide
Each eluent was continuously pumped through the column, and the resulting fractions were analyzed to determine the chromium concentration in the eluate and calculate the chromium recovery efficiency.
Chromium Recovery Results
The results showed that regeneration with alkaline sodium chloride solutions and ammonium chloride solutions containing ammonia provided the most effective chromium recovery.
Using sodium chloride solutions containing sodium hydroxide resulted in chromium recovery efficiencies of up to 99.7%, with chromium concentrations in the recovered solution reaching approximately 19 g/L. Ammonium chloride solutions with ammonia achieved recovery efficiencies of approximately 98–99%, with chromium concentrations of 14–23 g/L in the eluate.
In contrast, regeneration using alkaline sodium sulfate solutions was less effective, achieving chromium recovery efficiencies of 78–96% and significantly lower chromium concentrations in the recovered solution.
These results demonstrated that certain regeneration solutions can efficiently recover chromium while maintaining the adsorption capacity of the ion exchange resin for repeated treatment cycles.
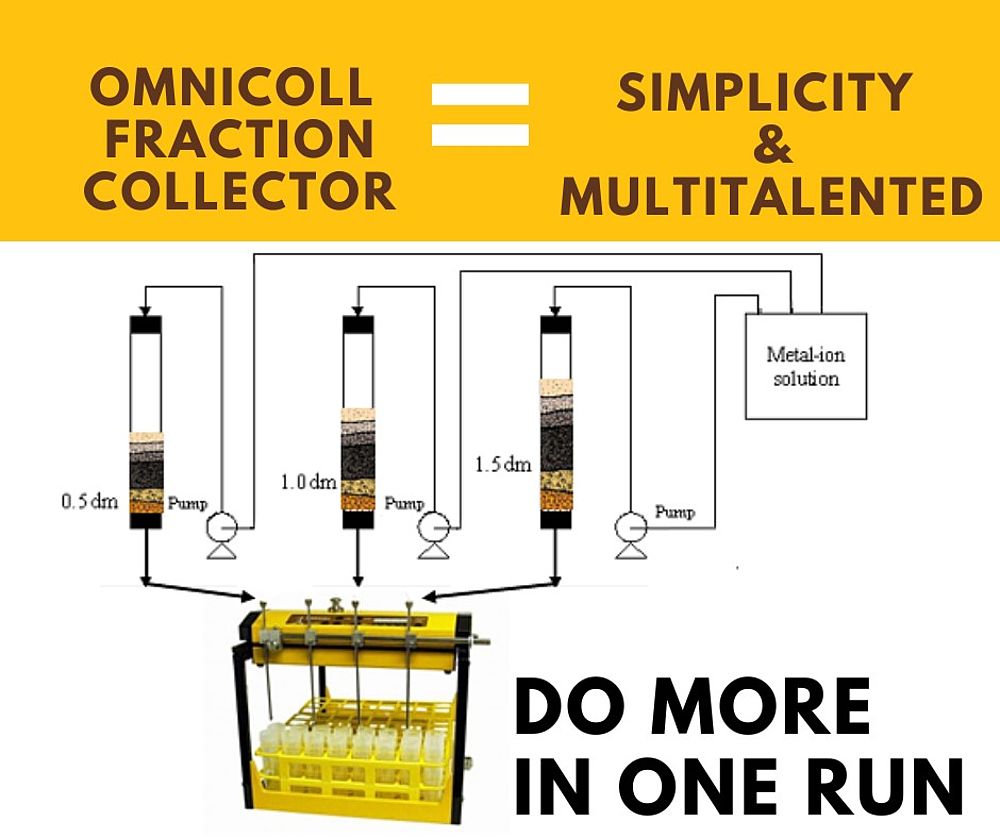
Parallel multiple Column purification with LAMBDA OMNICOLL Multichannel Fraction Collector
The LAMBDA OMNICOLL multichannel fraction collector enables parallel multiple column purification, allowing simultaneous operation and fraction collection from multiple chromatography or ion-exchange columns within a single system. Using LAMBDA multichannel accessories, different solvents, solutes, and column types can be processed in parallel, significantly reducing experimental time while maintaining high precision and reproducibility.
This multiple column fraction collection system ensures consistent, automated sampling across all channels, supporting accurate monitoring of adsorption, breakthrough, and desorption processes. By increasing throughput and enabling direct comparison of multiple conditions, the OMNICOLL enhances efficiency in high-throughput chromatography, ion-exchange studies, and advanced separation workflows.
Discover how automated fraction collection can enhance your separation experiments.
For more information, contact us at sales@lambda-instruments.com
More information find in the product specific page at https://www.fractioncollector.info/multi-stream-fraction-collection/